ISOPROPANOL 99,9% (technical spirit), L
4.50 €
2-propanol, CAS 67-63-0, isopropanol, INCI ISOPROPYL ALCOHOL, isopropyl alcohol, isopropyl alcohol, technical spirit, defoamer, cosmetic alcohol
Parameter | Attribute |
Isopropanol | 2-propanol, isopropanol, isopropyl alcohol |
Formula | C3H8O |
Structure | 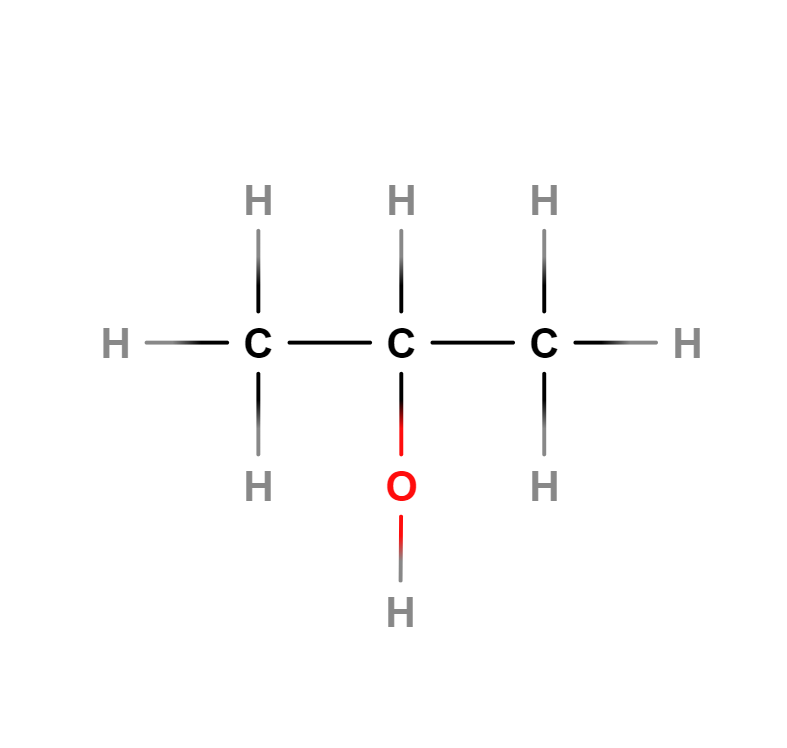  |
IUPAC | Propan-2-iol |
INCI | ISOPROPYL ALCOHOL |
CAS | 67-63-0 |
Molar mass | 60,1 g/mol |
Density | 0,786 g/cm3 (20 °C) |
Solubility | Miscible with water, benzene, chloroform, ethanol, ether, glycerin. Soluble in acetone. |
Also called isopropyl alcohol. It is a colourless, flammable compound with a strong odour and the chemical formula C3H8O.
It is used in the manufacture of a wide range of industrial and household chemicals and is a common ingredient in chemicals such as antiseptics, disinfectants and detergents.
Solvent. Isopropyl alcohol dissolves many non-polar compounds. It also evaporates quickly and leaves virtually no trace of grease compared to ethanol and is relatively non-toxic compared to alternative solvents. It is therefore widely used as a solvent and as a cleaning fluid, especially for fat dissolution.
Examples of applications of the material include cleaning of electronic devices such as contact pins (e.g. in ROM cartridges), magnetic tape and disc heads (such as audio-video recorders and floppy disk drives), laser lenses (e.g. on CDs, DVDs) in optical disc devices, and the removal of thermal paste from heatsinks and IC packages (e.g. CPUs).
In cosmetics, isopropyl alcohol is one of the most commonly used alcohols. It is used as an antiseptic in cosmetics: it is found, for example, in aftershave lotions, hand lotions, hand sanitisers, perfumes, etc. Main INCI functions:
- Antifoaming: eliminates foaming during manufacturing / reduces foaming in liquid end products
- Solvent: dissolves other substances
- Viscosity control: increases or decreases the viscosity of cosmetics
- Perfume: Used for perfumes and aromatic raw materials
Intermediate material for the production of other materials. Isopropyl alcohol is esterified to produce isopropyl acetate, another solvent, which is reacted with carbon disulphide and sodium hydroxide to produce sodium isopropyl xanthate, a herbicide and ore flotation reagent. Isopropyl alcohol reacts with titanium tetrachloride and aluminium to form titanium and aluminium isopropoxides, a catalyst and chemical reagent respectively.
In medicine. Used as a direct hand disinfectant (75 % aqueous solution) or for the production of disinfectant wipes (wipes are moistened with a 60-70 % aqueous solution of isopropanol). Isopropyl alcohol is used as a water-drying agent to prevent otitis externa.
For cars. Isopropanol is one of the main additives for "gas drying" fuels. With alcohol, the water in the fuel does not stratify and freeze, but is instead mixed in the fuel. Can be sold as an aerosol as an ice melting agent for windscreens and door locks. Isopropyl alcohol is also used to remove traces of brake fluid from hydraulic brake systems to prevent brake fluid (usually DOT 3, DOT 4 or mineral oil) from contaminating the brake pads, which would impair the vehicle's braking.
In the laboratory. May be a non-toxic alternative to formaldehyde and other synthetic preservatives for living tissues. For this purpose, 70 to 99 % isopropanol solutions are used. Isopropyl alcohol is often used in DNA extraction by adding it to the DNA solution. The addition of isopropanol to the DNA solution causes the DNA to precipitate and can be separated by centrifugation. DNA is insoluble in isopropanol.
Important: Add the item to your basket, fill in the recipient's details and confirm your order. Thank you!
To save your precious time, we will deliver your order to your address at a time convenient for You!
*- The pictures of the goods may not correspond to the actual appearance, colour, assembly or shape of the goods and their packaging. The information in the product description is of a general nature and may not correspond to the information on the packaging of the product and may not be the exact use of the product. The information given on the stocks and prices of goods may, in certain cases, differ from the actual prices and stocks of goods
Signal word: DANGER |
Hazard icons:
|
Danger phrases: H225: Highly flammable liquid and vapour H319: Causes severe eye irritation H336: May cause drowsiness or dizziness |
Precautionary statements: P210: Keep away from heat sources/sparks/open flames/hot surfaces. - Do not smoke. P233: Keep container tightly closed. P243: Take precautions to avoid static discharge. P305 + P351 + P338: In case of contact with eyes: Wash gently with water for several minutes. Remove contact lenses, if present and if easy to do so. Continue to wash eyes. P337 + P313: If eye irritation persists: seek medical attention. P403 + P235: Store in a well-ventilated place. Keep in a cool place |
Related products
(8 other products in the same category)












