|
Parameter |
Attribute |
||||
|
Citric acid |
Citric acid, food grade citric acid, E330, citric acid monohydrate |
||||
|
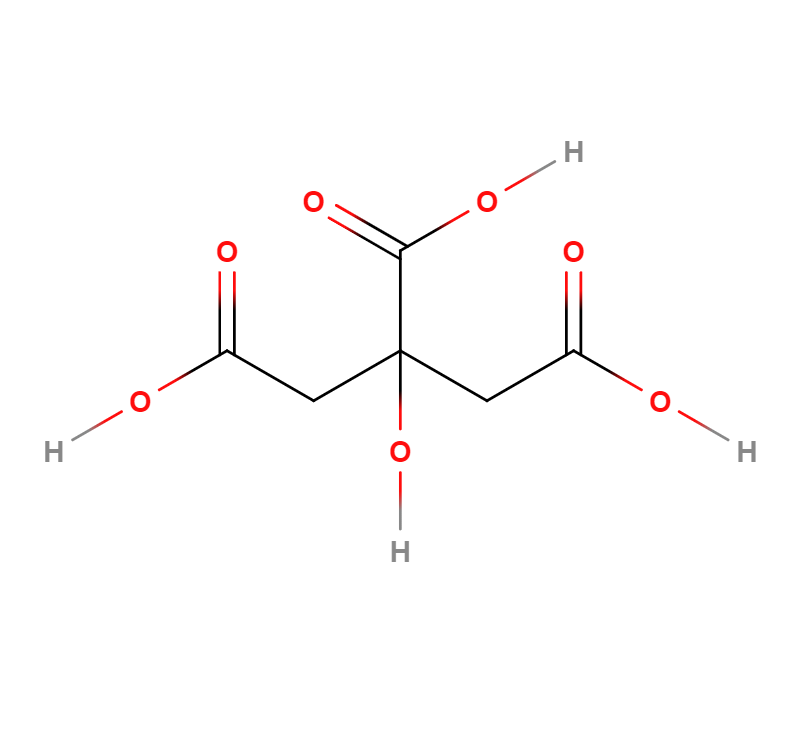
Formula |
C6H8O7 |
||||
|
Structure |
|
||||
|
IUPAC |
2-Hydroxypropane-1,2,3-tricarboxylic acid |
||||
|
INCI |
CITRIC ACID |
||||
|
CAS |
77-92-9 |
||||
|
Molar mass |
Monohidrate – 210,038 g/mol |
||||
|
Density |
Monohidrate– 1,542 g/cm3 |
||||
|
Solubility |
In water: 117.43 g/100 mL (10 °C) |
In ethanol: 62 g/100 g (25 °C) |
In amilacetate: 4.41 g/100 g (25 °C) |
In diethyl ether: 1.05 g/100 g (25 °C) |
In 1,4-dioksane: 35.9 g/100 g (25 °C) |
In food and drink industry, citric acid is used as a spice, flavoring additive or preservative. In the EU, citric acid additives in food products are labelled with an E indicator - E330. In food supplements, citric acid is used in the form of citrates (salts) of various metals. In ice cream, citric acid is used as an emulsifier to prevent the fat from separating from the whole mass, and in caramel to prevent the formation of sucrose (sugar) crystals on the surface of the caramel (inhibits the crystallisation of sucrose). Citric acid is used as a complement to sodium bicarbonate. In cooking it can be used instead of vinegar or lemon juice. Can be used in food coloring to balance the pH value.
In agriculture, it is used to regulate the acidity of water. At a pH of around 5.5, the uptake of all trace elements is highest. This means that less fertiliser is needed, absorption is easier, and plant growth is faster and yields higher. When using citric acid to acidify water, the hardness of the water must be known. The most common application rates are 100-500g of citric acid per 1000L of water. The exact amount of acid is selected either by field trials or by recalculation based on water quality parameters.
|
Acid |
Concentration |
g/m3 neutralisation of water with 50 ppm alkalinity |
ml/m3 neutralisation of water with 50 mg/L (ppm) alkalinity |
Received element |
|
Citric acid |
99.9 |
133.34 |
205.61 |
- |
In cleaning industry, as a means or agent (additive) for the formation of complex compounds. Citric acid forms compounds with metals to make them soluble. Effectively cleans and prevents the formation of new lime deposits in water evaporators, boilers, kettles etc. It is very commonly used in coffee machine descalers, both liquid (as a component) and solid (both pure and as a co-ingredient). Citric acid can be used to soften water by binding metals in hard water, allowing soaps and detergents to foam effectively. A 6% citric acid solution effectively cleans hard water stains on glassware. Can be used in shampoos to remove waxes from hair.
For regeneration of the charge of water filters, citric acid is used to remove iron, calcium, magnesium and other metals from the anion exchange charge resin of water softening filters. The quantity used is 0,5-1 kg citric acid per bag of salt tablets.
In industry, citric acid is used to dissolve rust on steel or as a substitute for nitric acid to passivate stainless steel.
In cosmetics, citric acid is used in almost all cosmetic products as an ingredient that regulates the pH value. It can be used as a skin and facial care product to remove dead skin cells and accelerate the formation of new ones. Citric acid is suitable for the production of face masks:
- For a pouty complexion, a mask of citric acid and milk can be used: in a bowl, mix ¼ cup of water, ¼ cup of low-fat milk and 2 tablespoons of citric acid powder. Apply the well-mixed mixture to the face and leave for 15 minutes. When the mask has hardened and started to crumble, wash your face with lukewarm water.
- Mask for unclogged pores Mix ½ cup water, 2 tbsp citric acid powder, ½ cup orange juice and a packet of gelatine without additives in a saucepan. Heat and stir the mixture over medium heat until all the ingredients are well dissolved. Allow the mixture to cool and then place in the refrigerator for 25 minutes. It can then be used as a face mask by applying it to the face, letting it sit for 30 minutes until it hardens, then gently removing the mask from the face.
Its functions (INCI):
- pH regulator : Stabilises the pH of cosmetics
- Chelating : Reacts and forms complexes with metal ions that could affect the stability and / or appearance of cosmetic products
- Masking : Reduces or inhibits the odor or basic taste of the product
In medicine. Prevents the formation of kidney stones by binding calcium. It is a good antioxidant which neutralises free radicals that promote the formation of cancer cells (tumours). Helps the body to better absorb minerals from foods. Can cure a sore throat by killing the bacteria that cause infection by rinsing the throat with an aqueous solution of citric acid.
Other uses of citric acid: it can be used as an odorless alternative to white vinegar in acid dyes. The citric acid salt sodium citrate is a component of the Benedict's reagent, which is used to identify and quantify reducing sugars. Citric acid is used in photography for the production of photographic film. Citric acid itself, as well as potassium and sodium citrates, can be used to regulate blood acidity. With other additives, it can be used as a bath soap, salt - when reacted with water it produces a foaming effect. A 10% pharmaceutical grade citric acid solution can be used as a carpet cleaner to remove stains.
Important: Add the item to your basket, fill in the recipient's details and confirm your order. Thank you!
To save your precious time, we will deliver your order to your address at a time convenient for You!
*- The pictures of the goods may not correspond to the actual appearance, color, assembly or shape of the goods and their packaging. The information in the product description is of a general nature and may not correspond to the information on the packaging of the product and may not be the exact use of the product. The information given on the stocks and prices of goods may, in certain cases, differ from the actual prices and stocks of goods
**- The product complies with the requirements for food additive E330 but is not intended for use as a food additive.
|
Signal word: Caution |
|
Hazard icons:
|
|
Danger phrases: H319 Causes severe eye irritation |
|
Precautionary statements: P264 Wash hands thoroughly after use. P280 Wear eye/face protection. P305 + P351 + P338 IN CASE OF CONTACT WITH EYES: Wash gently with water for several minutes. Remove contact lenses, if present and if easy to do so. Continue to wash eyes. P337 + P313 If eye irritation persists: seek medical advice. P391 Collect leaking material. |
Related products
(8 other products in the same category)